
Hospitals and the Spread of Infection
Even the strictest hospital manual cleaning protocols cannot prevent the spread of infections. Studies show that up to 50%2 of patient environments may remain infected after a routine cleaning. Microorganisms can remain present in the air for up to seven days,3 making these unclean spaces a hazard for patients, caregivers, and families.
2.Peters, A., Otter, J., Moldovan, A. et al. Keeping hospitals clean and safe without breaking the bank; summary of the Healthcare Cleaning Forum 2018. Antimicrob Resist Infect Control 7, 132 (2018). https://doi.org/10.1186/s13756-018-0420-3 3. Fernstrom A., Goldblatt M., “Aerobiology and Its Role in the Transmission of Infectious Diseases.” Journal of Pathogens vol.2013.
Innovative Solutions
Cleanis’ award-winning CareBag® solution is a first-to-market innovation that simplifies the unpleasant task of patient waste management and helps decrease the spread of infection. In fact, in 2020, the French Department of Health officially recommended that hospitals use the CareBag® solution to stem the spread of COVID-19.1
1.METHODOLOGY GUIDE : “PREPARATION AGAINST THE COVID-19 EPIDEMIC: Healthcare facilities, ambulatory medicine and medico-social facilities”. Ministry of Solidarity and Health of the French Republic (February 20th, 2020).
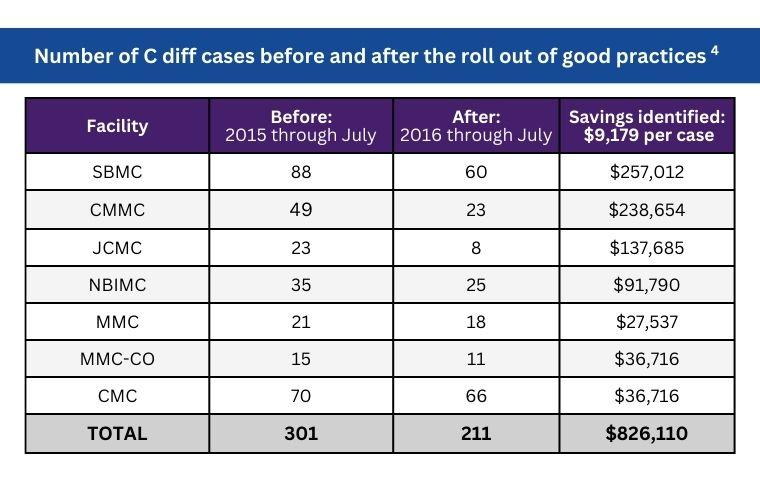
Proven Results
Multiple clinical studies have found that using CareBag®, combined with other best hygiene practices, is a proven method of reducing the spread of infection. According to a clinical study of a seven-hospital system, the CareBag® solution helped reduce C. diff by 30%.4
4. Yaney, Eileen MS, CIC; Filippone, Christine DNP, MSN, CIC, et all. ”Clostridium difficile Reduction Challenges in a Multi-System Hospital System.” RWJ Barnabas Health. Poster. 2016.
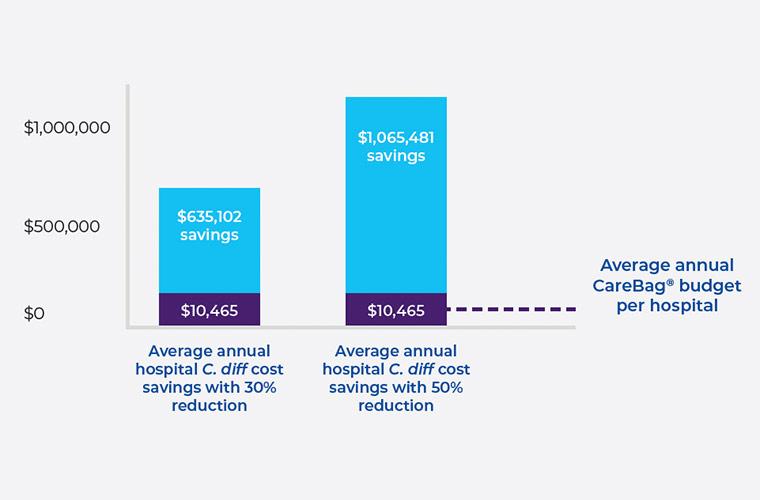
Cost Savings
The average hospital has 63 7,8 healthcare-associated C. diff cases per year at a cost of $34,157 9 per case. The estimated annual cost per facility is $2,151,891.
A small investment in CareBag® products saves the average hospital between $635k and $1.065M every year.10
7.Jarvis, William R; Schlosser, JoAnn; et. al. “National point prevalence of Clostridium difficile in US health care facility inpatients, 2008.” American Journal of Infection Control. 2009 May;37(4):263-70. 8. 2021 AHA Statistics. American Hospital Association. 9. Joint Commission Center for Transforming Healthcare. “Reducing Clostridium Difficile Infections.” From a comprehensive study of data from 2005 to 2015 published on the National Institutes of Health website. Zhang, Shanshan; Palazuelos-Munoz, Sarah, et al. “Cost of hospital management of Clostridium difficile infection in United States—a meta-analysis and modelling study.” Infect Dis. Published online 2016 Aug 25. 10.Cleanis calculations based upon average CareBag® hospital budget and cost of C. diff and hospital reported C. diff reductions.

Nurse Approved
Over 600 hospital CareBag® users overwhelmingly said that Cleanis®’ flagship solution helped improve their workday.5
According to a clinical study, even the number of steps required to care for patient waste is reduced with the use of CareBag®. The number of steps required for caring for an infectious patient was reduced from 20 to nine.6

